Perspectives
Rheumatoid arthritis: Giving our members the treatment they need, Part 1
Treatment to target: measuring disease activity and adjusting therapy
December 27, 2017Prime’s analysis of integrated medical and pharmacy data gives us insights into whether members are getting the most effective treatment for rheumatoid arthritis (RA), according to clinical guidelines. Following clinical guidelines improves the standard of care. Part 1: What is RA and why is it important? Part 2: The role of csDMARDS and b/tsDMARDS in treating RA. Part 3: What can a PBM and plan sponsor do to help?
Rheumatoid arthritis (RA) is a complex autoimmune disease causing joint pain and stiffness in both large and small joints. It eventually causes permanent damage to joints. It affects about 150,000 of Prime’s 20 million members–three times more women than men. This is a prevalence of about 0.7 percent of commercially insured adult members aged 18-65. This agrees with other published studies, which means that about 1.3 million adults in the United States aged 18-65, have RA.
Thirty years ago, with limited treatment options, an RA diagnosis often meant a painful, crippling decline and a shortened life expectancy. Today, due to advances in therapy, more people with RA live independently without major disability.1,2 Important advances have included:
- Use of methotrexate as a disease-modifying anti-rheumatic drug (DMARD)
- Initiation of drug therapy soon after diagnosis rather than treating symptoms with pain medications or much less effective drugs such as gold salts.
- Clinical evidence supporting methotrexate as the best first-line therapy for RA and as a part of therapy for individuals who require treatment with additional agents.
- Development of a long list of biologic DMARDs and a new category of non-biologic DMARDs that together comprise the leading expense category of specialty drugs.
The challenges?
- Diagnosing RA promptly and starting treatment right away
- Finding the right combination of drugs for each member with RA (this may take years of trying many combinations)
- Documenting the progression of the RA, adjusting and adhering to therapy (which requires good communication between the member and the health care team (supported by a specialty pharmacy)
This series of articles will illustrate how we are performing on these challenges, based on our analysis of integrated medical and pharmacy claims. It will show opportunities for interventions to improve performance to improve the standard of care.
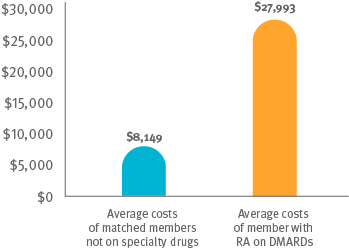
Cost comparison between members on DMARDs and matched members not on specialty drugs
Why is it important to focus on RA?
Treating RA correctly keeps people healthy and active with their families and in their communities. It keeps them working longer. Treating RA is expensive. It’s a big investment. Members being treated with DMARDs have three times the medical and pharmacy costs as members who are not being treated with these specialty drugs for RA.3,4
We need to maximize that investment to achieve the best health outcomes.
How do you treat RA?
The symptoms of RA can fluctuate and flare over time. A drug may stop working or become less effective. A treatment plan may need to change or evolve with the progression of the disease.
Finding a treatment protocol that works can be a process of trial and error:
- Trying different drugs
- Trying different doses of different drugs
- Different combinations of drugs
- It can take years to get a treatment regimen that relieves symptoms and improves the quality of life for a member with RA
Leaders in the field agree that treatment to target — by measuring disease activity and adjusting therapy accordingly — optimizes outcomes in RA.
How do you measure an ache? What is “treatment to target?”
“Treatment to target” for a member with RA is the same idea as using a hemoglobin A1C to adjust diabetes medication treatment. A doctor uses a quantitative disease measure to establish a base level score for the member with RA, such as:5, 6, 7
Disease Activity Score-28 (DAS28-CRP and DAS28-ESR) or Simplified Disease Activity Index (SDAI-CRP)
Each of these creates a score from a combination of:
- Measures from various lab tests
- Counts of the number of swollen joints
- Patient inventory: Member with RA answers an inventory of questions about life activities and physical abilities
- Doctor inventory: Doctor observations
This index is revisited at every visit to the doctor. At each appointment, the member and the doctor set a new goal. Changes in the score become part of the evaluation of the member’s progress. The following are treatment to target recommendations for ongoing monitoring of RA.8
When first diagnosed and when the RA is active, the member should see the doctor every three months to monitor and adjust the treatment plan.
If treatment has been successful, the lab results and the index will help to confirm that disease progression has slowed. When tests demonstrate that damage to joints has stopped (inactive RA), the member with RA and doctor may see each other less frequently – perhaps every six months.
If the RA remains inactive, the member may continue to see the doctor every six months. They monitor the treatment plan together. They may discuss tapering down medicine. This may not happen for years, if at all.
The next article in this series looks at the drugs that treat RA.
References
- Fleischmann R. “Don’t forget traditional DMARDs: Old friends are still useful.” Rheumatology(2011);50:429-430.
- Jurgens MS, et al. “The use of conventional disease-modifying anti-rheumatic drugs in established RA.” Best Pract Res Clin Rheumatol (2011);25:523-533.
- Bowen KL, Gleason PP. Rheumatoid arthritis 2016 prevalence, drug treatment, and total medical and pharmacy claims expense in a 15 million member commercially insured population. Poster presentation, AMCP 2017. J Manag Care Pharm 2017:23(10-a):S74. Accessed at: https://www.primetherapeutics.com/content/dam/corporate/Documents/Newsroom/Pressreleases/2017/document-amcpposter2017-ra.pdf
- Bowen K, Gleason PP. Prevalence and Cost of Autoimmune Specialty Drug Use by Indication in a 4.4 Million Member Commercially Insured Population Continuously Enrolled 4 Years, 2012-2015. Poster presentation, AMCP 2016. J Manag Care Pharm 2016:22(10-a):S83
- Bowen KL, Gleason PP. Incidence Rate of Biologic/Targeted Synthetic (b/ts)Disease Modifying Antirheumatic Drugs (DMARDs) for Rheumatoid Arthritis (RA), Preceding Therapy and Time to Discontinuation in a Commercially Insured Population. Poster presentation AMCP October 2017. AMCP 2017. J Manag Care Pharm 2017:23(10-a):S73. Accessed at: https://www.primetherapeutics.com/content/dam/corporate/Documents/Newsroom/Pressreleases/2017/document-amcpposter2017-radmards.pdf
- Smolen JS, Breedveld FC, Burmester GR, et al. Treating rheumatoid arthritis to target: 2014 update of the recommendations of an international task force. Ann Rheum Dis 2016;75:3-15.
- Smolen JS, Aletaha D, McInnes IB, et al. Rheumatoid arthritis. Lancet 2016;388:2023-2038.
- Solomon DH, Bitton A, Katz JN, et al. Review: treat to target in rheumatoid arthritis: fact, fiction, or hypothesis? Arthritis Rheumatol 2014;66:775-782.
Related news
Perspectives
July 25, 2024
Quarterly Drug Pipeline: July 2024
Clinical insights and competitive intelligence on anticipated drugs in development
Perspectives
July 22, 2024
Oncology Insights: 2024 ASCO Annual Meeting key findings
Findings from this year’s American Society of Clinical Oncology (ASCO) Annual Meeting will likely lead to clinical practice changes and U.S. Food and Drug Administration (FDA) drug approvals or expansions
Perspectives
July 16, 2024
LISTEN NOW: Beyond the business – Stories of corporate kindness | Pharmacy Friends Podcast
In this episode, we talk about how our employees' help goes beyond our work in health care, aiding in philanthropic efforts