Uncovering the true cost of your pharmacy benefit
Comparing the cost of pharmacy benefit managers (PBMs) can be a challenge. That’s because not all PBMs start with the same net cost before subtracting discounts and rebates. What’s more, they may present it all in complex, confusing spreadsheets. The good news is there is a single number you can look at when comparing PBMs: your net cost per member per month, also known as PMPM.
TRANSPARENT SAVINGS
Consistently delivering low PMPM

*Than national benchmarks
COMPREHENSIVE STRATEGY
Employing multiple drug management methods
The most effective PBMs help deliver the best patient outcomes at a lower cost. Prime takes a holistic approach to help ensure our formularies include the best options, promote evidence-based biosimilars and offer industry-leading utilization programs.

Formulary management
Some PBMs prioritize high-priced drugs over lower-cost options due to manufacturer rebates. But Prime focuses on the lowest-cost, most clinically effective options to keep costs low and improve health outcomes.

Specialty drug management
Prime is focused on improving access to low-cost specialty drugs by developing innovative products and driving the use of evidence-based biosimilars.
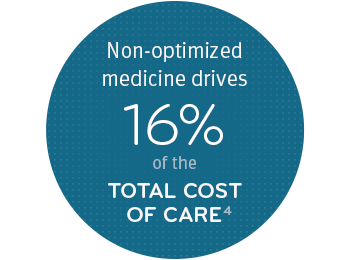
Utilization management (UM)
We help our clients choose UM programs that not only provide their members safe, cost-effective medicines, but also address industry-wide problems like inappropriate use and fraud, waste and abuse.
Related news
Perspectives
July 25, 2024
Quarterly Drug Pipeline: July 2024
Clinical insights and competitive intelligence on anticipated drugs in development
Perspectives
July 22, 2024
Oncology Insights: 2024 ASCO Annual Meeting key findings
Findings from this year’s American Society of Clinical Oncology (ASCO) Annual Meeting will likely lead to clinical practice changes and U.S. Food and Drug Administration (FDA) drug approvals or expansions
Perspectives
July 16, 2024
LISTEN NOW: Beyond the business – Stories of corporate kindness | Pharmacy Friends Podcast
In this episode, we talk about how our employees’ help goes beyond our work in health care, aiding in philanthropic efforts
1 Milliman survey, 2020; commercial business including fully insured and self-insured
2 Prime Therapeutics internal data, 2017; savings based on actual savings reports, inclusive of all formulary savings
3 U.S. Food and Drug Administration, and Federal Trade Commission. (2020, February 3). FDA and FTC announce new efforts to further deter anti-competitive business practices, support competitive market for biological products to help Americans [Press release]. https://www.fda.gov/news-events/press-announcements/fda-and-ftc-announce-new-efforts-further-deter-anti-competitive-business-practices-support
4 Watanabe, J. H., McInnis, T., & Hirsch, J. D. (2018, March 26). Cost of prescription drug-related morbidity and mortality. Annals of Pharmacotherapy, 52(9). https://journals.sagepub.com/doi/10.1177/1060028018765159