PerspectivesStories,
Prime gets drug manufacturers to put more than product on the line
Ultra high-cost drugs are changing the face of value-based contracting.
November 25, 2019We started off ahead of the game
Value-based contracts have only been around for about ten years.1 Prime was an early adopter. Prime’s integration with our health plans made us a good partner for early value-based contracts. We signed our first value-based contracts back in 2011.
Value-based contracts were originally designed for chronic lifetime therapies for larger populations. We look to understand long-term value and short-term costs. The contracts could include health monitoring and testing. We could look at adherence over time. We had a longer time horizon to track outcomes. We had history and a larger population to forecast utilization and financial impacts. (Notice the elements of time here.)
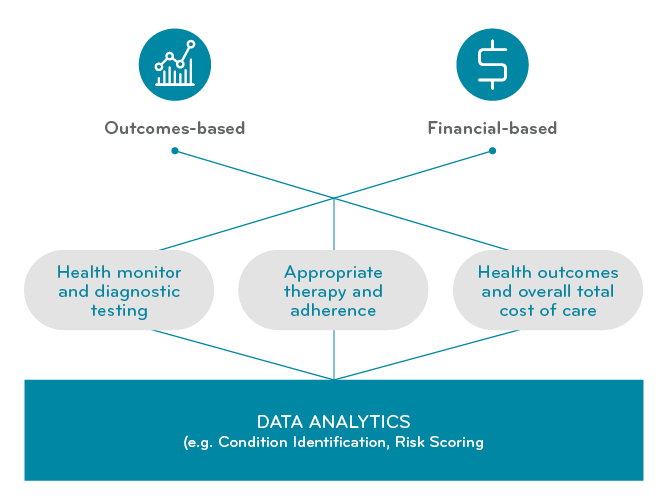 Value-based arrangements are different for gene and one-time therapies
Value-based arrangements are different for gene and one-time therapies
With today’s new ultra-high cost one-time treatments, the calculations look different. Many of the treatments are one-time (we think), but no one really knows yet what’s going to happen five and ten years out. Long-term safety data is not yet available. There are no large-scale clinical trials. The value based contract has to rely more heavily on health outcomes and total cost of care.
Price-to-value models become more important in crafting the value-based contracts for high cost drugs
I represent Prime as a member of the Institute for Clinical and Economic Review (ICER). Prime uses ICER’s value framework to assess the cost of drugs from a value perspective. The framework calculates a drug’s value per QALY (quality-adjusted life year). In its most basic form, QALY stands for one year in good health. In the United States, ICER uses a range of $50,000 to $150,000 to quantify the value of that one year of good health, which seems like a fair investment because the median household income is $50,000 a year in the U.S. Several factors go in Several factors go into a thoughtful QALY calculation. ICER has a helpful report on these calculations.
 Does the drug extend the person’s life span? Can the person go back to work? Does it make the person more independent? Does it help the person walk again that couldn’t before? Does it prevent future hospitalizations? to a thoughtful QALY calculation. ICER has a helpful report on these calculations.
Does the drug extend the person’s life span? Can the person go back to work? Does it make the person more independent? Does it help the person walk again that couldn’t before? Does it prevent future hospitalizations? to a thoughtful QALY calculation. ICER has a helpful report on these calculations.
Knowing a drug’s value framework can identify a price to value gap – the difference between its value-based price and the actual drug price. That can give us a place from which to negotiate with a drug manufacturer for a value-based contract. Sometimes we can close those gaps.
Get ready by launch
With high-cost drugs, it’s important to have a value-based contract ready as soon as possible. We proactively monitor the drug pipeline and carefully prepare clinical recommendations and pricing strategies even before the launch of a high cost drug. We conduct pre-launch conversations with drug manufacturers about prices and contracts.
Prime currently has value-based contracts with manufacturers on several ultra-high cost therapies to manage orphan conditions, such as those seen in infants and young children.
 Contracts will continue to evolve
Contracts will continue to evolve
Additional expectations for value-based arrangements for high cost drugs will include:
- Capturing outcomes not available through claims
- Tracking members over extended periods of time
- Ensuring that diagnostic tests are performed and results reported
- Custom and non-traditional reporting
- Specified coverage determinations and reimbursement
- Administering rebates/financial guarantees
- Audit capabilities
- Best price implications of guarantees
Most gene and one-time therapy treatments will occur at specialized centers. This means that most patients (and their families) will travel for treatment. This could cause wide variability in cost and reimbursement rates. Contract details will need to accommodate multi-state structure and process.
These drugs have already begun to shape the future of health care. As Prime leads our Blue Plan partners toward that future, we will leverage our unique business model and analytic capabilities to execute value-based contracts for ultra-high cost drugs.
Related news
Perspectives
July 25, 2024
Quarterly Drug Pipeline: July 2024
Clinical insights and competitive intelligence on anticipated drugs in development
Stories
July 22, 2024
Introducing the latest issue of our medical and pharmacy benefit management report
The summer 2024 issue of the Magellan Rx Report provides insights on the industry’s most groundbreaking managed care solutions in some of the most complex areas of health care
Perspectives
July 22, 2024
Oncology Insights: 2024 ASCO Annual Meeting key findings
Findings from this year’s American Society of Clinical Oncology (ASCO) Annual Meeting will likely lead to clinical practice changes and U.S. Food and Drug Administration (FDA) drug approvals or expansions
 Contracts will continue to evolve
Contracts will continue to evolve