Perspectives
Keytruda continues to stack up FDA approvals for more cancer indications
Merck’s cancer treatment is a golden goose: delivering more than $4.5 billion in sales in 2021
January 26, 2022Drug name: Keytruda (pembrolizumab)
Manufacturer: Merck
Keytruda is approved for the treatment of:
- Several kinds of skin cancer (melanoma, head and neck squamous cell cancer, hepatocellular carcinoma, Merkel cell carcinoma, cutaneous squamous cell carcinoma)
- Classical Hodgkin Lymphoma and a type of non-Hodgkin’s Lymphoma called primary mediastinal large B-cell lymphoma
- Small cell lung cancer and non-small cell lung cancer
- Cancers in the esophagus and the lining of the stomach
- Cancers in the cervix and the uterus
- Cancers in the urinary tract, bladder, and in the kidneys
- An aggressive form of breast cancer called triple-negative breast cancer (TNBC)
- Colon and rectal cancer
- Various forms of cancer cell mutations that impact other cancer growth
Overview of Keytruda (pembrolizumab)
Pembrolizumab is a monoclonal antibody that changes the patient’s immune system to help control the growth of cancer cells. The application of this type of antibody can result in a long-lasting response and can extend survival in patients with a wide range of cancers.2 For some cancers, this drug is recommended following use of other treatments. For other cancers, it can be a first line treatment.
Pembrolizumab can cause the immune system to attack normal organs and tissues in any area of the body and can affect the way they work. These problems can sometimes become severe or life-threatening and can lead to death.
Patients being treated with pembrolizumab will be monitored closely for symptoms and signs that may be clinical manifestations of underlying immune-mediated adverse reactions. This will include checking liver enzymes, creatinine and thyroid functions at baseline and during treatment.
Patients with TNBC treated with pembrolizumab in the neoadjuvant setting, will have their blood cortisol monitored at baseline, prior to surgery, and as clinically indicated.
Definition: neoadjuvant therapy is delivered before surgery with the goal of shrinking a tumor or stopping the spread of cancer to make surgery less invasive and more effective.
In cases of suspected immune-mediated adverse reactions, a physician orders additional tests to exclude other causes, like infection, and begins medical management as appropriate.
Pembrolizumab is administered after dilution as an intravenously (IV) infusion over 30 minutes in a hospital or outpatient setting with health care supervision. Preparation of pembrolizumab is the same regardless of fixed or weight-based dosing. It is usually administered every three or six weeks. Keytruda has an annual list price of $175,000.3
Keytruda sales by quarter, 3Q 2014 to 3Q 20213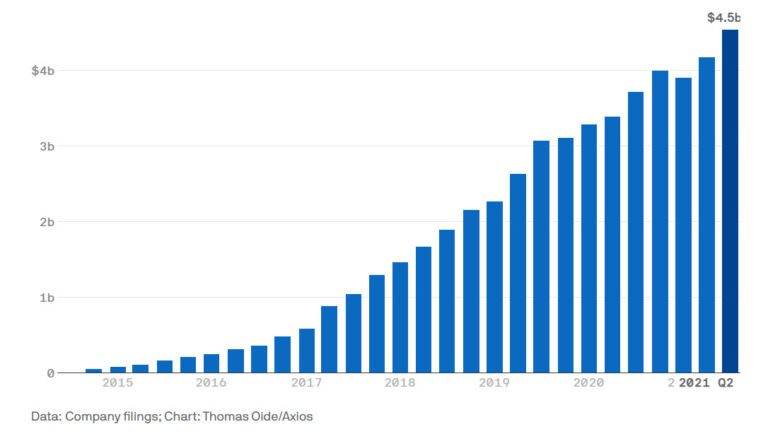
Nasdaq places Keytruda as the third top selling drug in the world, after Pfizer’s vaccine drug and Abbvie’s Humira® (adalimumab).4
Benefit: Medical benefit
Prime monitors the drug pipeline5
The drug pipeline is full of new, groundbreaking specialty drugs that may help members live better and feel well. Prime focuses on clinical strategies designed to keep clients ahead of drug trends — because it’s easier to manage change when you see it coming.
References
- label (fda.gov) Accessed at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2021/125514s096lbl.pdf
- Akinleye, A., Rasool, Z. Immune checkpoint inhibitors of PD-L1 as cancer therapeutics. J Hematol Oncol12, 92 (2019). https://doi.org/10.1186/s13045-019-0779-5 Accessed at: https://rdcu.be/cFn1A
- Merck’s cancer drug Keytruda on pace for $17 billion of sales – Axios. Accessed at: https://www.axios.com/keytruda-sales-merck-drug-prices-b1d81138-5e88-43fa-bbb5-d074ecee713b.html
- Top 10 Blockbuster Drugs In 2021, Dec, 23, 2021. RTTNews.com © 2022 Nasdaq, Inc. Accessed at: https://www.nasdaq.com/articles/top-10-blockbuster-drugs-in-2021
Related news
Perspectives
July 25, 2024
Quarterly Drug Pipeline: July 2024
Clinical insights and competitive intelligence on anticipated drugs in development
Perspectives
July 22, 2024
Oncology Insights: 2024 ASCO Annual Meeting key findings
Findings from this year’s American Society of Clinical Oncology (ASCO) Annual Meeting will likely lead to clinical practice changes and U.S. Food and Drug Administration (FDA) drug approvals or expansions
Perspectives
July 16, 2024
LISTEN NOW: Beyond the business – Stories of corporate kindness | Pharmacy Friends Podcast
In this episode, we talk about how our employees' help goes beyond our work in health care, aiding in philanthropic efforts