Perspectives
Biosimilar strategy for bevacizumab using MedDrive™
Preferring two biosimilars can save plan sponsors tens of millions of dollars
November 30, 2021Avastin® (bevacizumab) from Genentech was first approved in 2004. It has received FDA approval for treatment of:1
- advanced colon cancer (2004)
- advanced lung cancer (2006)
- kidney (2009) cancer and
- brain (glioblastoma) cancer (2009)
Bevacizumab is also used off-label to treat age-related wet macular degeneration, where it is given by injection into the eye (intravitreal). (Biosimilar recommendations for that use will be covered in another article.)
Bevacizumab is a monoclonal antibody that inhibits the development of new blood vessels. Since tumors rely on blood vessels to get the nutrients they need to survive, this drug fights cancer by preventing the formation of blood vessels that feed the cancer tumor.2
Definition: angiogenesis – the development of new blood vessels.
Definition: vascular endothelial growth factor (VEGF) is a protein that promotes the growth of new blood vessels. VEGF is a key requirement for tumor growth.2
Because Bevacizumab inhibits VEGF, an angiogenesis growth factor, it is sometimes referred to as a VEGF or angiogenesis inhibitor.2
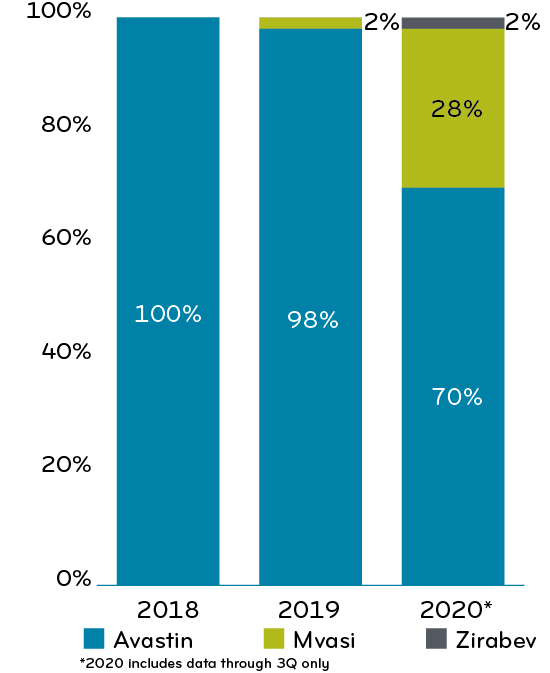
In 2021, Bevacizumab cost Prime’s clients $1.05 per member per month (PMPM); these drug costs were incurred on the medical side. Avastin, as a reference drug for bevacizumab, has two biosimilars: Mvasi, launched in 2019, and Zirabev, launched in 2020. The introduction of biosimilars provided some price relief. Bevacizumab has been growing at a rate of 4.87 since 2018.
Changes in percentage spend across bevacizumab products, 2018-20203
The growth rate comes from new patients. The pool of people getting various types of cancer grows every day — 7 to 16% of the patients that take bevacizumab are new every month.
Bevacizumab is generally administered intravenously by a health care professional every 14 days. Dosages are weight based and timing of cycles may vary. Depending on the cancer it is treating, it is often given in combination with other chemotherapy drugs. While not a cure, bevacizumab can extend survival time for advanced cancers.
The reference drug annual wholesale acquisition cost (WAC) for the reference drug is about $144,000. The annual WAC for the biosimilars is about 25% less than that.4
Biosimilars are beginning to move the dial
There are several studies that look at the price-to-value cost effectiveness of bevacizumab for the various cancer indications it can treat. In general, results show improvements in overall survival in terms of months for advanced/late stage cancers, but costs vary widely. Bevacizumab studies do not easily fit into regular price-to-value analyses because of their use in treating late stage cancers.6-8
Mvasi launched mid-year 2019; by the end of 2019, Mvasi filled 13.5% of the bevacizumab prescriptions.
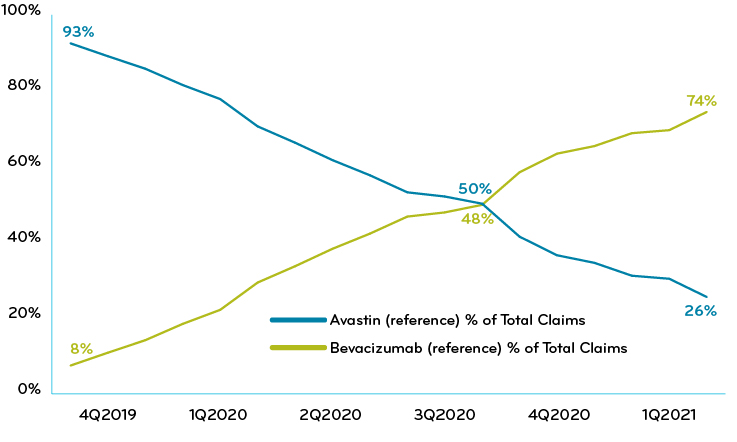
In 2020, Prime’s overall book of business has used a mix of strategies to manage this drug. About half of the plans treated the reference drug and biosimilars the same. The other half had some form of preferred biosimilar strategy. Overall, the movement to biosimilars steadily rose to about 74% (Chart 1).
Chart 1: Prime’s commercial book of business: % of claims reference drug vs biosimilar
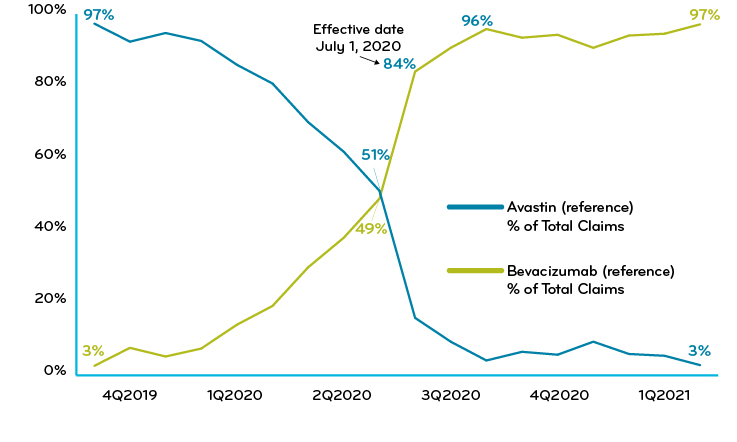
One of Prime’s Blue Plan clients implemented a utilization management policy requiring a trial of the biosimilar Zirabev prior to the use of the reference drug Avastin or the biosimilar Mvasi. Effective date was 7/1/2020. Within six months after adopting the strategy, the client achieved total biosimilar market share greater than 97% and saw $0.04 PMPM in savings (Chart 2).
Chart 2: Blue Plan implementation of UM policy: % of claims for reference vs biosimilar drugs
MedDrive strategy for bevacizumab: prefer two biosimilars
The launch of MedDrive signaled that Prime and its Blue Plan clients were going to get more aggressive about biosimilar savings. Bevacizumab is one example of that strategy in action.
Prime’s recommended MedDrive strategy for bevacizumab is for a combination of two preferred biosimilars over the more expensive reference drug. Prime’s Blue Plan clients are now in the process of transitioning to preferring two biosimilars over the reference drug, Avastin.
Five more bevacizumab biosimilars are in the pipeline: four with 2021 anticipated approval dates and one anticipated in 2022.9 This kind of competition will drive prices down further, increasing potential for savings.
Now that Prime has a made its MedDrive biosimilar recommendation to clients, we anticipate biosimilar market share to increase and savings to build.
MedDrive medical solutions is a complete toolkit
MedDrive leverages the collective strength of Prime’s client membership to help control medical costs. Client savings are obtained through:
- Improved rebates from manufacturers,
- Lower net costs with shift of use from expensive medical drugs to lower cost alternatives, such as biosimilars
- Lower cost alternatives within the same therapeutic class
Prime’s Blue Plan clients are executing preferred biosimilar programs are designed to save them hundreds of millions of dollars. We’ll continue to showcase these drugs and this analysis in coming weeks. For more information, contact your local Prime representative.
References
- FDA Avastin Approval History, updated by Judith Stewart, BPharm on May 5, 2021. © 2000-2021 Drugs.com. Accessed at: https://www.drugs.com/history/avastin.html
- Angiogenesis inhibitors-National Cancer Institute. U.S. Department of Health and Human Services. USA.gov. Accessed at: https://www.cancer.gov/about-cancer/treatment/types/immunotherapy/angiogenesis-inhibitors-fact-sheet.
- Prime book of business data.
- Yang, J., Liu, R., Ektare, V., Stephens, J., & Shelbaya, A. (2021). Does Biosimilar Bevacizumab Offer Affordable Treatment Options for Cancer Patients in the USA? A Budget Impact Analysis from US Commercial and Medicare Payer Perspectives. Applied health economics and health policy, 19(4), 605–618. https://doi.org/10.1007/s40258-021-00637-5.
- Minion LE, Bai J, Monk BJ, Robin Keller L, Ramez EN, Forde GK, Chan JK, Tewari KS. A Markov model to evaluate cost-effectiveness of antiangiogenesis therapy using bevacizumab in advanced cervical cancer. Gynecol Oncol. 2015 Jun;137(3):490-6. doi: 10.1016/j.ygyno.2015.02.027. Epub 2015 Mar 10. PMID: 25766118. Accessed at: https://pubmed.ncbi.nlm.nih.gov/25766118/.
- “Close Call for Cost-Effectiveness of Bevacizumab in Ovarian Cancer,” by John Chan, MD. Oncology & Biotech News, June 2012, Volume 6, Issue 6. Accessed at: https://www.onclive.com/view/close-call-for-cost-effectiveness-of-bevacizumab-in-ovarian-cancer.
- Klag, N., Walter, A. C., Sheely, K. M., Manahan, K. J., & Geisler, J. P. (2016). Is the routine use of bevacizumab in the treatment of women with advanced or recurrent cancer of the cervix sustainable?. ClinicoEconomics and outcomes research: CEOR, 8, 287–291. https://doi.org/10.2147/CEOR.S71218.
- Chen, Z., Zhan, M., Tian, F., & Xu, T. (2020). Cost‑effectiveness analysis of the addition of bevacizumab to temozolomide therapy for the treatment of unresected glioblastoma. Oncology Letters, 19, 424-430. https://doi.org/10.3892/ol.2019.11099.
- Drug Pipeline Quarterly Update: September 2021 (primetherapeutics.com)
Related news
Perspectives
July 25, 2024
Quarterly Drug Pipeline: July 2024
Clinical insights and competitive intelligence on anticipated drugs in development
Perspectives
July 22, 2024
Oncology Insights: 2024 ASCO Annual Meeting key findings
Findings from this year’s American Society of Clinical Oncology (ASCO) Annual Meeting will likely lead to clinical practice changes and U.S. Food and Drug Administration (FDA) drug approvals or expansions
Perspectives
July 16, 2024
LISTEN NOW: Beyond the business – Stories of corporate kindness | Pharmacy Friends Podcast
In this episode, we talk about how our employees' help goes beyond our work in health care, aiding in philanthropic efforts