Perspectives
Research on Botox use for migraines shows big drop off rate
But hang on: migraine sufferers may now benefit from a new class of therapy.
April 22, 2019Migraines are surprisingly prevalent. They affect 39 million people in the United States.1
Most migraine sufferers experience attacks once or twice a month, but more than 4 million people have chronic daily migraines. That means they have at least 15 migraine days per month; 90 percent of them are unable to work or function normally during those days.1
The good news is that there are several prescription drugs from several different classes that treat acute migraines. Many of those drugs have generics available. Plus, there are new drugs to try and still others in the pipeline. But it can be a real headache for migraine patients to keep trying different drugs, hoping to find one that will give them relief.
Prime’s study on Botox for migraines found high costs and waste
Most of the time, patients who try Botox have not responded to other conventional migraine therapies. Most of those are generic drugs, taken by mouth. Botox is a biologic therapy. It’s more expensive and requires a health care professional to administer.
The study identified more than 59,000 claims for Botox® (onabotulinumToxinA) from October 2017 to September 2018 among 15 million commercially insured members. Botox for migraines accounted for 65 percent of the category spend. We wanted to look specifically at use of Botox for migraine. A total of 2,473 members met study requirements for continuous enrollment and were identified as new to Botox therapy. For these members, the study showed:
- Annual cost of Botox for chronic migraine use was more than $6,300 when used according to manufacturer label
- Drug waste and provider administration accounted for 34 percent of costs
- Less than half of new Botox users received the four recommended treatments during the first year of therapy
- Use of Botox did not decrease migraine-related medical or pharmacy costs; rather it was associated with a three-fold increase in total migraine-related costs.
A closer look at real-world use tells an interesting story
FDA-approved dosing for Botox is 155 units per treatment. However, vials sizes come in 100 units and 200 units. That causes waste of 45 units per treatment, for which providers are allowed to bill.
Botox treatment for chronic migraines is quarterly. Each treatment involves 31 separate injections of 5 units each in seven areas around the face and neck. It can take up to six months to see the maximum benefit from Botox.2 Prime’s study showed 18 percent of members do not receive the second treatment; another 18 percent drop before the third treatment. Only 47 percent of members in the study stayed adherent for the full year of the study.
The Botox treatment was not associated with a decline in migraine-related medical and pharmacy costs. Annual average migraine-related costs for the member before starting Botox migraine therapy was $2,467. In the year after starting Botox, the annual average migraine-related cost was $7,422.
Comparing Prime’s data with ICER’s evaluation
Prime uses the Institute for Clinical and Economic Review (ICER) and other cost-effectiveness information in its formulary decision-making process. Prime works to ensure that clinically effective, low net cost products are available. ICER reports evaluate the clinical and economic value of prescription drugs and assess price to value. ICER measures drug pricing against the value added in quality adjusted life years (QALY). Is the drug priced to the value it gives the patient?
ICER compared Botox and cGRPs
Much of ICER’s migraine treatment evaluation focused on a relatively new drug class called calcitonin gene-related peptide (cGRP) monoclonal antibodies. Three cGRP treatments have launched in the last year, with more in the pipeline. In ICER’s evaluation, Botox and the cGRP drug class appeared to have similar effectiveness with Botox reported to have lower costs resulting in Botox having a better QALY.3 In contrast, Prime’s real-world claims data and pricing experience shows that the cGRP drug class gives a distinct price to value advantage over Botox; by extension, cGRPs show a better QALY cost effectiveness than Botox using Prime’s real-world data inputs.4
Coverage of Migraine Treatments5
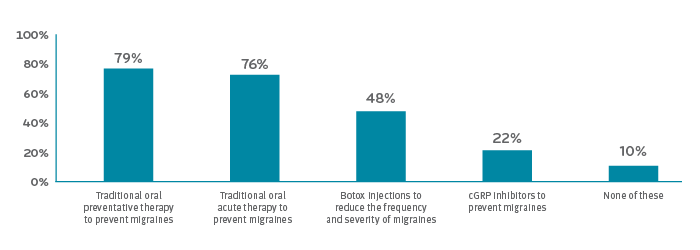
Health plans and employers are looking for guidance
In the Pharmacy Benefit Management Institute’s 2019 Trends in Specialty Drug Benefits, a few questions were added about coverage of migraine treatments. Most employers surveyed covered preventive and acute migraine drug therapy (79 percent and 76 percent respectively). Only half covered Botox therapy. Two thirds had step therapy requiring trial of at least one nonspecialty agent.5
Employers slowly becoming aware of cGRP therapy
Only 21 percent of employers know “a fair amount” about cGRP inhibitors.5 The rest either haven’t heard of them or have heard of them, but don’t know much. Seventy percent of employers weren’t sure whether they were going to add cGRPs to their formularies.5
Prime keeps clients on top of the pipeline
The PBMI research shows that employers want to cover proven migraine treatments, but don’t know enough about cGRPs yet.5 Prime can help here. Prime constantly monitors the specialty drug pipeline. By the time a new drug class like cGRP has been launched, we’ve studied the clinical data and conducted a full range of forecasting analyses. Our clients hear these reports in monthly pipeline webinars and read about them in weekly and monthly pipeline updates.
All our clients benefit from our expertise working with integrated medical and pharmacy claims data. We evaluate data from industry sources, but we test it against our real-world claims experience.
Prime’s research provides the evidence for recommendations
Based on available evidence, we expect similar safety and efficacy in treating chronic migraines from both cGRPs and Botox, although Botox has been on the market longer than cGRPs. Currently, cGRPs appear to be a lower net cost, price to value option for chronic migraine treatment.
At Prime’s quarterly Medical Business Committee (formerly known as the Specialty Drug Strategy Committee) meetings, we discuss these findings with pharmacy and medical directors from our Blue Plan clients. Together, we work towards ensuring access to the most cost-effective medications to improve health outcomes at the best price to value.
References
- About Migraine. © 2019 Migraine Research Foundation. Accessed at: https://migraineresearchfoundation.org/about-migraine/migraine-facts/
- What you need to know about Botox for Migraine. ©American Migraine Foundation. Accessed at: https://americanmigrainefoundation.org/resource-library/botox-for-migraine/
- The QALY: Rewarding the Care That Most Improves Patients’ Lives. Dec 2018. © Institute for Clinical and Economic Review. Accessed at: https://icer-review.org/wp-content/uploads/2018/12/QALY_evLYG_FINAL.pdf
- Prime internal data.
- Pharmacy Benefit Management Institute. 2019. Trends in Specialty Drug Benefits. Plano, TX. PBMI. Available from www. pbmi.com/Specialty Reports.
Drug names are the property of their respective owners.
Related news
Perspectives
April 23, 2024
Expert Clinical Network Insights: April 2024
A look into our Expert Clinical Network (ECN) – part of Prime/MRx’s value-based approach to medical and pharmacy benefit management that offers access to more than 175 national and world-renowned key opinion leaders in multiple disease categories who provide expertise on challenging prior authorization case reviews, peer-to-peer discussions, drug policy development and formulary guidance
Perspectives
April 23, 2024
April Fraud Focus – How to prioritize drug safety at home
While ensuring drug safety is often considered the responsibility of pharmaceutical industry players,…
Perspectives
April 19, 2024
AMCP 2024: Behind the Research with Prerak Parikh
Parikh, director of medical pharmacy strategy at Prime/MRx, shares the latest on interchangeable biosimilars