Perspectives
Biosimilar strategy for trastuzumab using MedDrive™
Two step program: One: Prefer biosimilars. Two: Save money
December 7, 2021Herceptin received its first FDA approval in 1998 for HER2+ metastatic breast cancer. In 2006, the FDA approved Herceptin to treat HER2+ early breast cancer. In 2010, the FDA approved Herceptin to treat HER2+ metastatic stomach cancer.1 It is often given as part of treatment along with a chemotherapy drug, or alone after treatment with multiple other therapies.
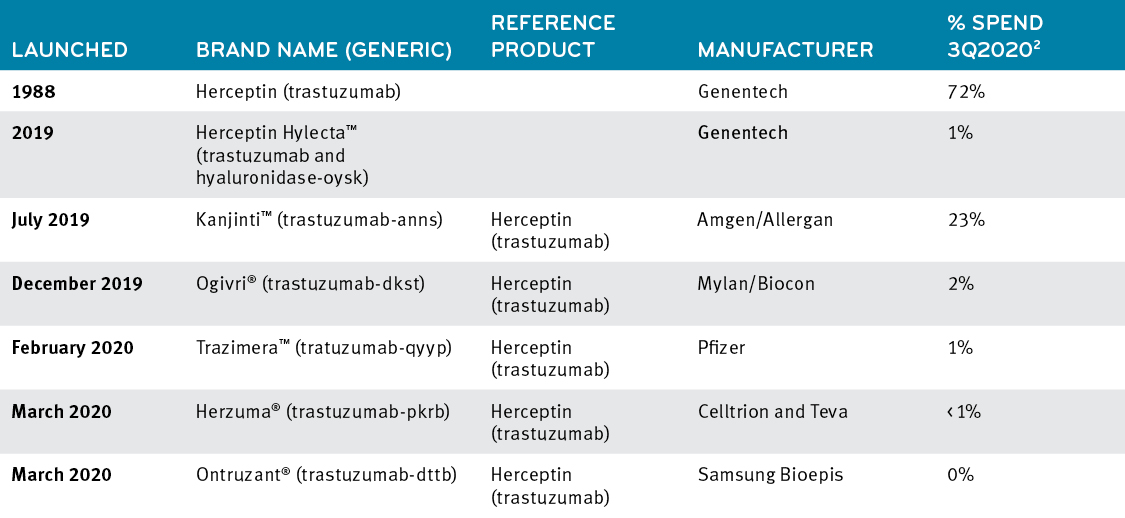
While the first biosimilar was launched in 2019, traction in the marketplace was slow until it was joined by other trastuzumab biosimilars in 2019 and 2020. Market share to begin to move, but primarily to Kanjinti, and only to 13.2%.
In 2020, biosimilars scraped their way to 29% market share. At that point, Prime had several Blue Plan clients preferring trastuzumab biosimilars.
Changes in percentage spend across trastuzumab products, 2018-20202
Genentech may have gotten a little boost in 2020 with a formulation of the drug (Herceptin Hylecta) that could be administered subcutaneously, instead of intravenously. This makes this formulation of trastuzumab faster and easier for patients who are familiar with it and tolerate it well. It may slow migration to the biosimilars.
Trastuzumab utilization has been shrinking at a 5.46% rate since 2018. Its cost on a per member per month basis is $1.10. Every month, about 7-10% of its utilizers are new starts.
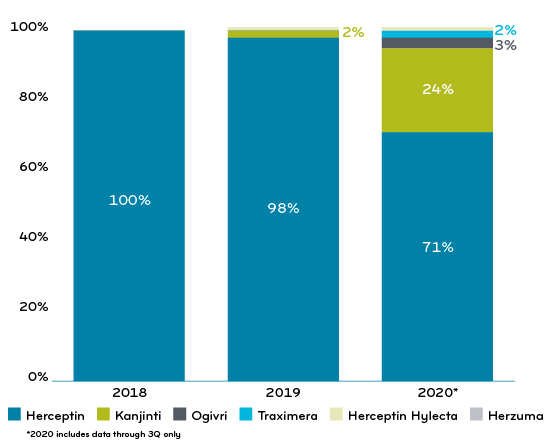
In 2020, about half of Prime’s Blue Plan clients were treating the reference drug and biosimilars at parity. The other half had some form of preferred biosimilar strategy. Chart 1 shows the result of that mixed approach on Prime’s book of business. It made a steady rise to a 73% biosimilar market share.
Chart 1: Prime’s commercial book of business: % of claims trastuzumab reference drug vs biosimilars
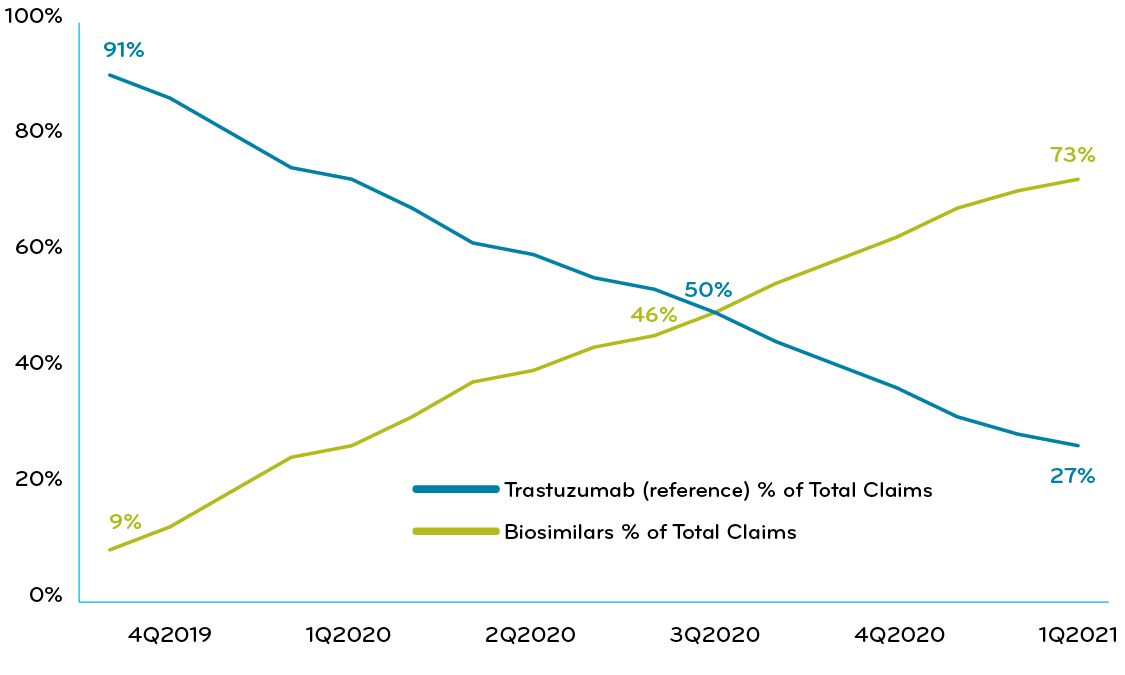
One Prime Blue Plan client implemented a utilization management policy requiring a trial of Trazimera (trastuzumab-qyyp) before use of the reference product or other nonpreferred trastuzumab agents. Within six months, the client achieved a biosimilar market share of greater than 95% and saw $0.18 PMPM in savings (Chart 2). This demonstrates the power of a preferred drug strategy.
Chart 2: A Blue Plan with UM policy requiring trial of Trazimera: % of claims Trazimera vs Herceptin reference drug
Trastuzumab is infused over a period of time in a doctor’s office, clinic or hospital. Depending on treatment schedule and dosing, it is given on a weekly schedule or an every three-week schedule until disease progression.
As they have come out, the prices of trastuzumab biosimilars have been 10 to 22% lower than the reference drug. A price-to-value review of trastuzumab suggests that that HER2+ early breast cancer therapy may be cost-effective in high-income countries.4 Perhaps some biosimilar competition within this drug class will help prices come down further.
Comparison of the wholesale acquisition cost (WAC) of Herceptin vs biosimilars in the United States as of Q2 2020 (in U.S. dollars)3
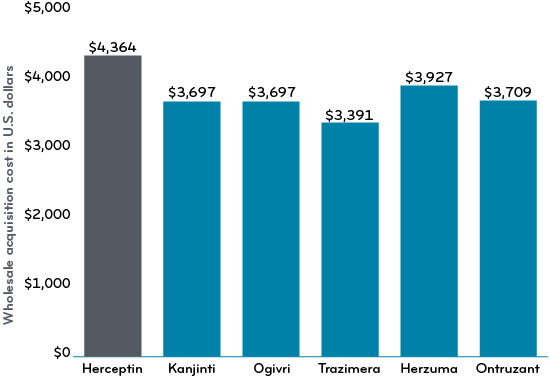
https://www.statista.com/statistics/1186607/comparison-wac-herceptin-vs-biosimilars-us/ U.S., 2nd quarter 2020
MedDrive strategy for trastuzumab: prefer two biosimilars
The launch of MedDrive (news release link) signaled that Prime and its Blue Plan clients were going to get more aggressive about biosimilar savings. Prime’s recommended MedDrive strategy for trastuzumab is for a combination of two preferred biosimilars ahead of the reference drug and the other biosimilars. Prime’s Blue Plan clients are now in the process of transitioning to preferring two biosimilars over the reference drug Herceptin.
At this time, there are no additional trastuzumab biosimilars in the pipeline.5
Now that Prime has a made its MedDrive biosimilar recommendation to clients, we anticipate biosimilar market share to increase and savings to build over the next year.
MedDrive medical solutions is a complete toolkit
MedDrive leverages the collective strength of Prime’s client membership to help control medical costs. Client savings are obtained through:
- Improved rebates from manufacturers
- Lower net costs with shift of use from expensive medical drugs to lower cost alternatives, such as biosimilars
- Lower cost alternatives within the same therapeutic class
Prime’s Blue Plan clients are executing preferred biosimilar programs that are designed to save them hundreds of millions of dollars. We’ll continue to showcase these drugs and this analysis in coming weeks. For more information, contact your local Prime representative.
References
- Biosimilar product information. U.S. Food & Drug Administration. Accessed at: https://www.fda.gov/drugs/biosimilars/biosimilar-product-information
- Prime book of business
- Comparison of the wholesale acquisition cost (WAC) of Herceptin vs biosimilars in the United States as of Q2 2020. Statistic.com. Accessed at: https://www.statista.com/statistics/1186607/comparison-wac-herceptin-vs-biosimilars-us/
Related news
Perspectives
April 23, 2024
Expert Clinical Network Insights: April 2024
A look into our Expert Clinical Network (ECN) – part of Prime/MRx’s value-based approach to medical and pharmacy benefit management that offers access to more than 175 national and world-renowned key opinion leaders in multiple disease categories who provide expertise on challenging prior authorization case reviews, peer-to-peer discussions, drug policy development and formulary guidance
Perspectives
April 23, 2024
April Fraud Focus – How to prioritize drug safety at home
While ensuring drug safety is often considered the responsibility of pharmaceutical industry players,…
Perspectives
April 19, 2024
AMCP 2024: Behind the Research with Prerak Parikh
Parikh, director of medical pharmacy strategy at Prime/MRx, shares the latest on interchangeable biosimilars