Perspectives
Biosimilar strategy for rituximab using MedDrive™
Going for tens of millions in annual savings, plan sponsors will prefer two biosimilars
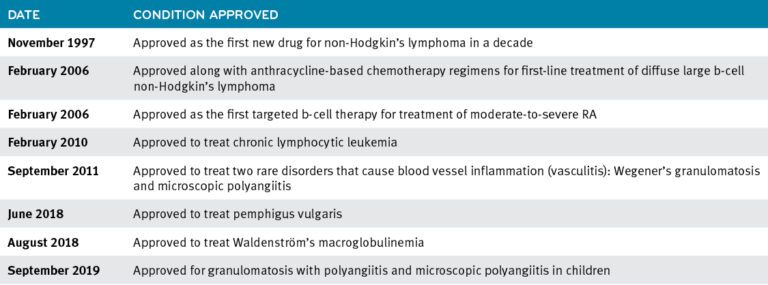
December 14, 2021Rituxan (rituximab) FDA-approved conditions1
In a healthy body, B-cells that are working correctly create antibodies that fight viruses and infections. In a body with cancer, B-cells forget that job and join the cancer cells.2,5
Monoclonal antibodies are a tool to shut down the B-cells of tumors. Rituximab is a manufactured monoclonal antibody that attaches to the CD20 receptor on the B-cells of tumors. It destroys them and prevents production of additional B-cells.3,5
The use of rituximab in treating rheumatoid arthritis (RA) is different. For a patient with RA, the B-cells in the immune system promote inflammation. When rituximab depletes the number of B-cells, it reduces that inflammation.4,5
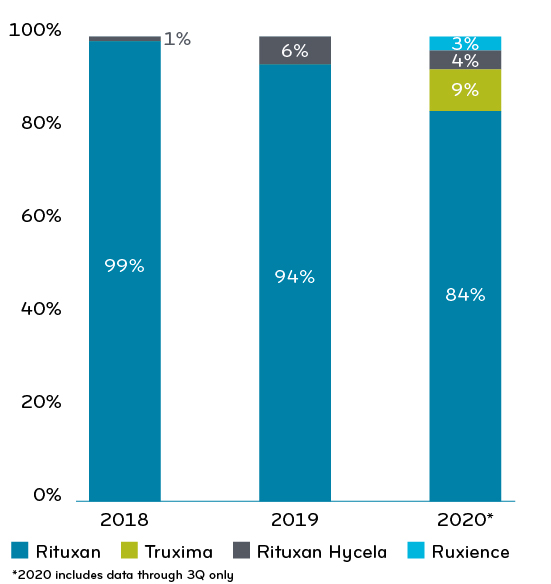
Changes in percentage spend across Rituximab products, 2018-20206
The reference drug got a boost in 2017 with a formulation of the drug (Rituxan Hycela) that could be administered subcutaneously instead of intravenously. That made use of rituximab much faster and easier for patients who were familiar with it and tolerated it well.2
This might have slowed rituximab’s migration away from Genentech, which produces both Rituxan and Rituxan Hycela. The biosimilars scraped together only 12% market share by 2020. Drug spend per member per month (PMPM) was $1.16. Half of that ($0.58) was for oncology.
Rituxan is administered as a slow intravenous infusion by a health care professional at a medical facility or infusion center. For some cancers, it’s taken along with another chemotherapy. For some, it is used as alone as a first line therapy.
For oncology use, most patients receive one dose every 28 days for around six to nine months. For non-oncology use (mostly RA), patients receive two doses spaced two weeks apart; then receive that two-dose series every six to 12 months depending on severity of indication.4
What that means is that there is an ongoing churn in the utilizers of this product. Many patients are on rituximab for only six months to one year of therapy. Every month, about 30-35% of its utilizers are new patients. But on an ongoing basis, rituximab has only been growing at a rate of 3.12% since 2018.
Rituximab is expensive. The reference drug annual wholesale acquisition cost is more than $60,000, depending on the diagnosis and dosage. And for many cancers, it is given in addition to other treatments, which affects its price to value. But several studies show it is cost effective at extending life for specific cancers.10-12
Biosimilar savings
When Truxima (rituximab-abbs) was launched in 2019, it was priced only 10% lower than the reference drug.4 When Ruxience (rituximab-pvvr) and Riabni (rituximab-arrx) were launched in 2020 and 2021, their price was lower, 24% less than the reference drug.7,8,9
There is some opportunity for site of care savings in the non-oncology areas, potentially more than $50 million. Because Rituximab can have significant side effects, and the potential for member disruption could be high, we would work closely with our Blue Plan clients on recommending strategies around site of care.
Improving health outcomes and lowering costs
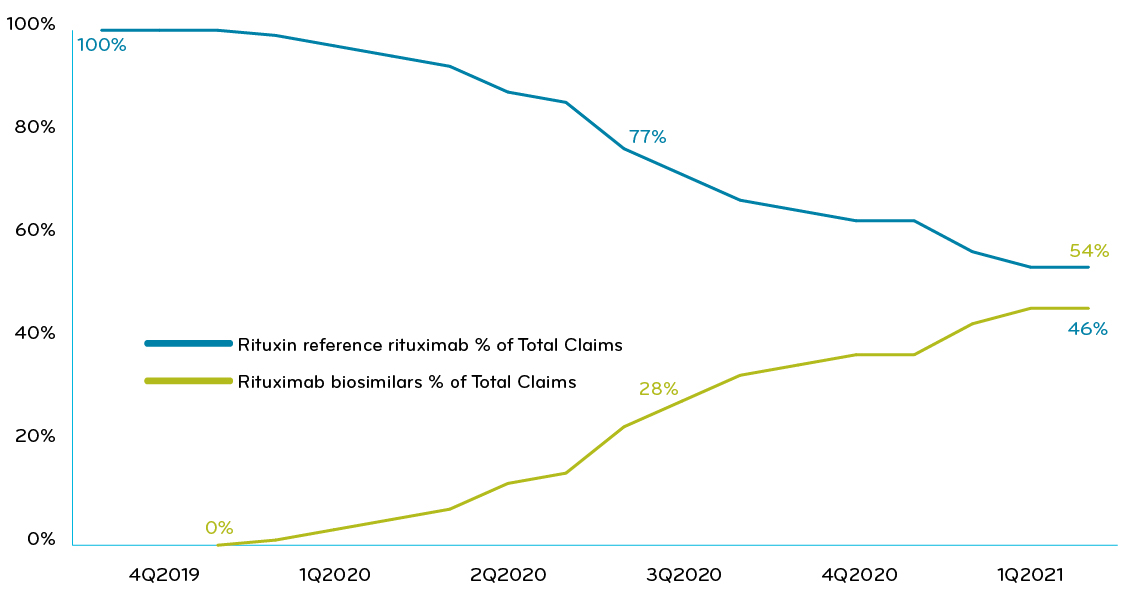
In 2020, Prime’s overall book of business has used a mix of strategies to manage this drug. About half of the plans treated the reference drug and biosimilars the same. The other half made the reference drug nonpreferred and gave one or both biosimilars preferred status. Overall, the movement to biosimilars has been slow (Chart 1). Improving from such a low biosimilar market share — 46% — the savings potential for rituximab is high. A lot of money is being left on the table.
Chart 1: Prime’s commercial book of business: % of claims of rituximab reference drug vs biosimilars, collectively
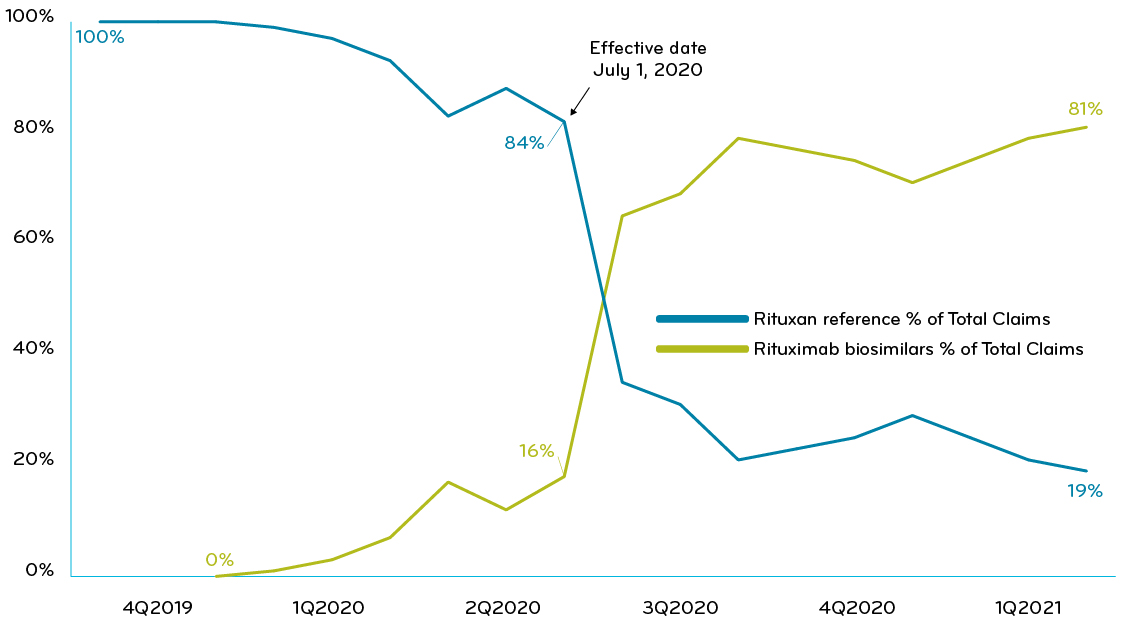
One of Prime’s Blue Plan clients executed a utilization management program that required trial of Ruxience (rituximab-pvvr) prior to use of nonpreferred rituximab drugs. Six months after adopting the strategy, the client achieved total biosimilar market share greater than 81% and saw $0.24 PMPM in savings (Chart 2).
Chart 2: A Blue Plan with UM policy requiring trial of Ruxience: % of claims of Ruxience vs other rituximab drugs
MedDrive strategy for rituximab: prefer two biosimilars
The launch of MedDrive (news release link) signaled that Prime and its Blue Plan clients were going to get more aggressive about biosimilar savings. Prime’s recommended MedDrive strategy for rituximab is for a combination of two preferred biosimilars over the reference drug and the other biosimilars. Prime’s Blue Plan clients are now in the process of transitioning to preferring two biosimilars over the reference drug Rituxan.
At this time, there are no additional rituximab biosimilars in the pipeline.13
Now that Prime has a made its MedDrive biosimilar recommendation to clients, we anticipate biosimilar market share to increase and savings to build over the next year.
MedDrive medical solutions is a complete toolkit
MedDrive leverages the collective strength of Prime’s client membership to help control medical costs. Client savings are obtained through:
- Improved rebates from manufacturers,
- Lower net costs with shift of use from expensive medical drugs to lower cost alternatives, such as biosimilars
- Lower cost alternatives within the same therapeutic class
Prime’s Blue Plan clients are executing preferred biosimilar programs are designed to save them hundreds of millions of dollars. We’ll continue to showcase these drugs and this analysis in coming weeks. For more information, contact your local Prime representative.
References
- Rituxan FDA Approval history, by Judith Stewart, BPharm. Drugs.com. Accessed at: https://www.drugs.com/history/rituxan.html
- Yuen, G. J., Demissie, E., & Pillai, S. (2016). B lymphocytes and cancer: a love-hate relationship. Trends in cancer, 2(12), 747–757. https://doi.org/10.1016/j.trecan.2016.10.010
- Biosimilar product information. U.S. Food & Drug Administration. Accessed at: https://www.fda.gov/drugs/biosimilars/biosimilar-product-information
- Lopez-Olivo M, Amezaga Urruela M, McGahan L, Pollono EN, Suarez-Almazor ME. Rituximab for rheumatoid arthritis. Cochrane Database of Systematic Reviews 2015, Issue 1. Art. No.: CD007356. DOI:10.1002/14651858.CD007356.pub2. Accessed at: https://www.cochrane.org/CD007356/MUSKEL_rituximab-for-rheumatoid-arthritis
- Monoclonal antibody drugs for cancer: How they work. By Mayo Clinic Staff. © 1998-2021 Mayo Foundation for Medical Education and Research (MFMER). Accessed at: https://www.mayoclinic.org/diseases-conditions/cancer/in-depth/monoclonal-antibody/art-20047808
- Prime internal analysis, commercial book of business.
- Teva launches rituximab biosimilar Truxima in US. Biosimilars News. 05-06-20. © 2021 Generics and Biosimilars Initiative (GaBI). Accessed at: https://www.gabionline.net/biosimilars/news/Teva-launches-rituximab-biosimilar-Truxima-in-US
- Pfizer hits its biosimilar stride, by Jacob Bell. BioPharma Drive. Jan. 27, 2020. © 2021 Industry Dive. Accessed at: https://www.biopharmadive.com/news/pfizer-hits-its-biosimilar-stride/571136/
- Amgen’s Biosimilar RIABNI™ (rituximab-arrx) Now Available. 02-03-2021. Michigan Society of Hematology and Oncology. Accessed at: https://associationdatabase.com/aws/MSHO/pt/sd/news_article/347603/_PARENT/layout_details/true
- Khor, Sara et al. “Real world costs and cost-effectiveness of Rituximab for diffuse large B-cell lymphoma patients: a population-based analysis.” BMC cancer vol. 14 586. 12 Aug. 2014, doi:10.1186/1471-2407-14-586. Accessed at: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4148552/
- Knight C, Hind D, Brewer N, Abbott V. Rituximab (MabThera) for aggressive non-Hodgkin’s lymphoma: systematic review and economic evaluation. Health Technol Assess. 2004 Sep;8(37):iii, ix-xi, 1-82. doi: 10.3310/hta8370. PMID: 15361313. Accessed at: https://pubmed.ncbi.nlm.nih.gov/15361313/
- Hornberger JC, Best JH. Cost utility in the United States of rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone for the treatment of elderly patients with diffuse large B-cell lymphoma. Cancer. 2005 Apr 15;103(8):1644-51. doi: 10.1002/cncr.20956. PMID: 15756658. Accessed at: https://pubmed.ncbi.nlm.nih.gov/15756658/
- Drug Pipeline Quarterly Update: September 2021 (primetherapeutics.com)
Related news
Perspectives
April 25, 2024
Drug Approvals Monthly Update: April 2024
This monthly update of United States (U.S.) Food and Drug Administration (FDA) approvals…
Perspectives
April 24, 2024
Prime/MRx resident wins AMCP Foundation Best Poster Award
Ai Quynh Nguyen, PharmD, was recently recognized for her research on opioid-prescribing patterns and outcomes
Perspectives
April 23, 2024
Expert Clinical Network Insights: April 2024
A look into our Expert Clinical Network (ECN) – part of Prime/MRx’s value-based approach to medical and pharmacy benefit management that offers access to more than 175 national and world-renowned key opinion leaders in multiple disease categories who provide expertise on challenging prior authorization case reviews, peer-to-peer discussions, drug policy development and formulary guidance